Pharma Labeling Compliance - Tips & Best Practices in 2025
Mar 31, 2025
Compliance with pharmaceutical labeling regulations is essential to avoid costly penalties, product recalls, and, more importantly, to protect public health.
In this article, we’ll explore key strategies and best practices for remaining compliant with pharma labeling compliance and how staying ahead of the game can improve both your operational efficiency and reputation.
Let’s begin!
What is Pharma Labeling Compliance, and Why Does It Matter?
Pharma labeling compliance refers to adhering to the rules and guidelines set by regulatory bodies like the FDA and EMA, ensuring that all labels on pharmaceutical products meet regulatory standards.
These labels provide essential information such as dosage, ingredients, warnings, and expiration dates, which are vital for patient safety.

In the pharmaceutical industry, labeling isn’t just about marketing; it’s about providing the correct and necessary information to healthcare professionals and consumers.
Ensuring compliance with labeling regulations helps prevent medication errors, misbranding, and fraud, all of which can have severe legal and ethical implications.
The Regulatory Importance of Pharma Labeling Compliance
Pharmaceutical companies are subject to some of the most stringent labeling requirements due to the sensitive nature of their products.
Regulatory bodies such as the FDA have strict guidelines for the labeling of pharmaceutical products, ensuring that labels are accurate, informative, and unambiguous.
These regulatory agencies ensure that pharmaceutical labels:
Are accurate and truthful: Labels must contain correct and comprehensive information about the drug’s composition, dosage, and risks.
Provide clear instructions: Labels must be unambiguous, offering precise guidance on usage, side effects, and contraindications.
Ensure patient safety: Proper labeling helps healthcare providers administer drugs safely and effectively to patients.
Minimize legal risks: Non-compliance can result in recalls, penalties, and lawsuits that could harm both a company’s reputation and public health.\
Pharma FDA Labeling Guidelines: Ensuring Accuracy and Transparency
The FDA sets comprehensive labeling standards to ensure that pharmaceutical labels provide accurate, clear, and necessary information.
Key aspects of the FDA labeling requirements include:
Inclusion of Active and Inactive Ingredients: FDA regulations require that all medication labels include both active and inactive ingredients (21 CFR 201.10). This ensures transparency and helps healthcare professionals and consumers make informed decisions about drug usage.
Clear Labeling Format (The Physicians’ Labeling Rule (PLR)): The PLR improves how healthcare practitioners access and use medication labels. It includes specific sections like the Highlights of Prescribing Information and a Table of Contents (Page 2), which enhances clarity and consistency in drug labeling (21 CFR 201.66).
Prominence of Required Label Statements: FDA regulations specify that certain statements, such as the name and place of business of the manufacturer (21 CFR 201.62), must be prominently displayed on labels. This ensures that critical information is easily accessible to consumers and healthcare professionals.
Recent Labeling Compliance Issues in Pharma
Dr. Reddy's Labeling Issues in March 2025: Dr. Reddy's issued a nationwide recall of Levetiracetam in 0.75% Sodium Chloride Injection 1,000 mg/100 mL in the U.S. due to mislabeling of the infusion bag.
Aurobindo Pharma USA Recall (2020): Aurobindo Pharma USA recalled 7,440 bottles of Ibuprofen oral suspension due to a labeling error. This incident highlights how labeling mistakes can lead to product recalls and safety concerns.
Endo Pharmaceuticals Recall: Endo Pharmaceuticals initiated a recall due to labeling inaccuracies regarding the dosage regimen of Robaxin 750 mg tablets. This discrepancy could have resulted in patients taking incorrect doses, jeopardizing their well-being.
FDA Recalls Due to Labeling Errors (2012-2023): Approximately 8% of all FDA recalls between 2012 and 2023 were attributed to labeling issues. This trend emphasizes the ongoing challenge of ensuring accurate labeling in pharmaceutical products.
Key Considerations for Pharmaceutical Labeling Compliance
Incorporating a robust approach to pharmaceutical labeling compliance is essential for minimizing risks such as product recalls, penalties, and patient safety issues.
Below are best practices to follow for maintaining compliance:
1. Regular Review of Regulatory Guidelines
➸ What it is:
Regularly reviewing the latest labeling guidelines issued by regulatory bodies like the FDA ensures that your labels comply with current standards.
➸ Why it's important:
Compliance is an ongoing process, and regulations can change frequently. Staying up-to-date helps avoid penalties and ensures your product labels align with the latest requirements.
➸ How to do it:
Set up periodic reviews within your regulatory affairs team by subscribing to updates from regulatory agencies and attending industry conferences or webinars to stay informed about changes.
Additionally, AI Labeling Compliance Software can streamline this process by automatically monitoring regulatory updates and providing real-time alerts when relevant changes occur.
Pro tip
To stay ahead of regulatory changes, leverage Signify's automated regulatory monitoring to monitor evolving regulations and standards that impact your products.

With real-time regulatory updates and clear, actionable insights, Signify helps you focus on the changes that matter most to your business, allowing you to adapt proactively before any changes are enforced.
2. Clear and Accurate Label Content
➸ What it is:
Ensuring all content on your pharmaceutical labels is clear, accurate, and backed by clinical data.
This includes mandatory information such as the drug name, active ingredients, dosage instructions, contraindications, and storage instructions.
➸ Why it's important:
Accurate and clear labeling is essential to ensure patient safety, correct administration, and proper storage of medications. Misleading or incomplete labels can result in patient harm or regulatory action.
➸ How to do it:
Implement processes that verify data accuracy at multiple stages of the labeling process.
For example, clinical data, input from medical professionals, and legal reviews can be used to validate label content before final approval.
Additionally, AI-driven Labeling Compliance Software can help by cross-checking labels against regulatory requirements and automatically flagging inconsistencies or missing information.
Pro tip
Signify allows you to validate product labeling with precision by automatically inspecting artwork, packaging, and labels against applicable regulatory requirements.
This helps identify compliance gaps early in the design process, reducing costly revisions and ensuring that your labels are compliant before they reach the market.
3. Automated Systems for Labeling Management
➸ What it is:
Utilizing automated tools to manage the extensive documentation and updates required for pharmaceutical labels.
➸ Why it's important:
Automation reduces human error, ensures consistency across product labels, and speeds up the update process, especially when regulatory changes occur.
➸ How to do it:
Invest in label management software that not only ensures labeling compliance but also provides additional features such as document control, regulatory tracking, and workflow automation to streamline your overall compliance processes.
Pro tip
Signify is an AI-powered compliance management platform that simplifies regulatory tracking, risk assessments, and product labeling validation across industries like food and beverage, personal care, household goods, and more.
With its AI-driven automation, Signify streamlines complex tasks, reduces compliance burdens, and ensures your processes remain precise and up-to-date with evolving global regulations.
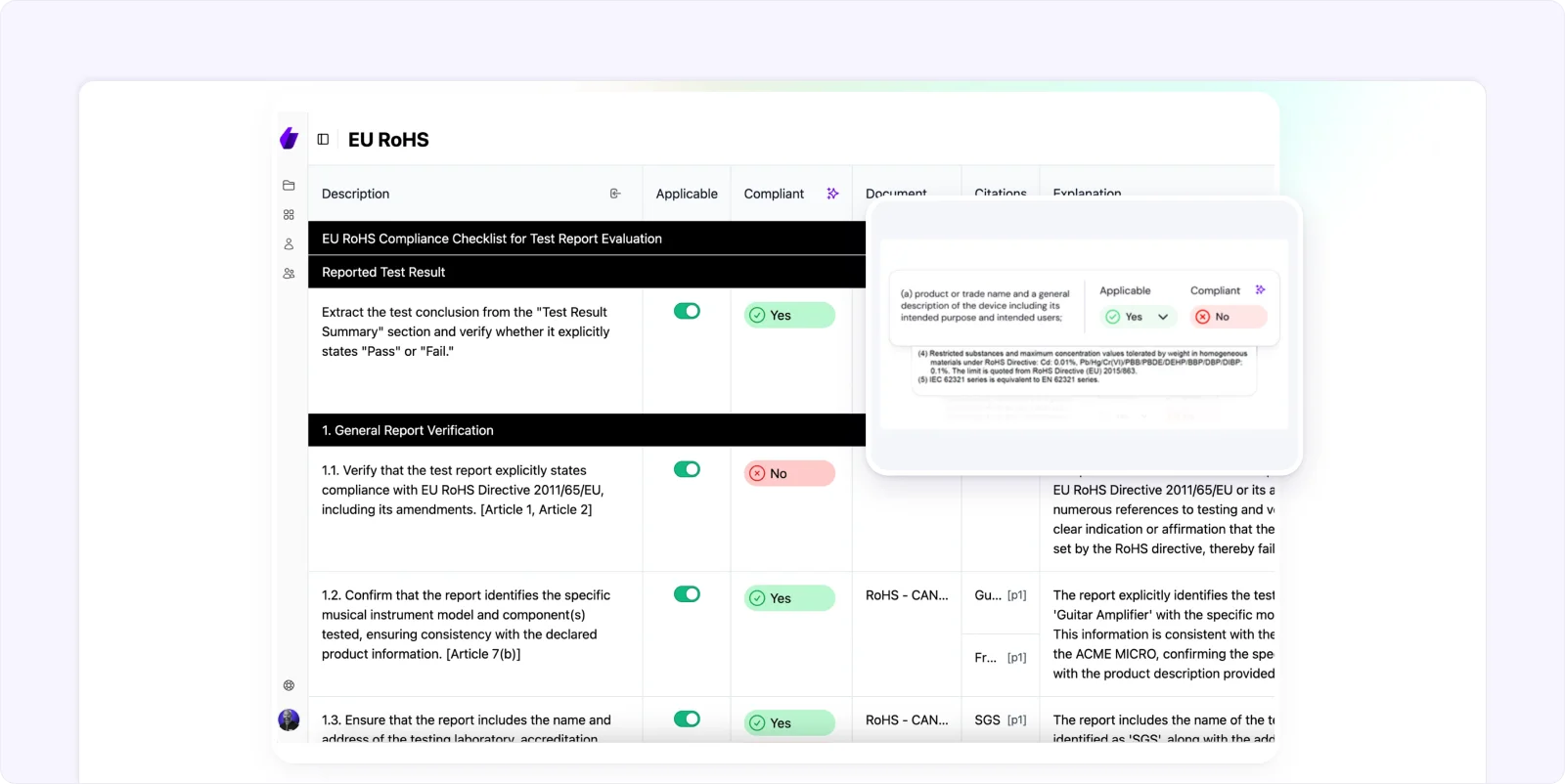
Whether you’re in manufacturing or quality assurance, Signify enhances operational transparency, allowing you to focus on delivering quality products.
4. Collaboration with Regulatory Bodies
➸ What it is:
Establishing a proactive relationship with regulatory agencies to stay informed about changes in labeling regulations and standards.
➸ Why it's important:
Open communication with regulatory bodies helps you understand the intent behind new guidelines and can help prevent costly mistakes in your labeling process.
➸ How to do it:
Designate a liaison between your company and the regulatory agencies.
This person or team should regularly communicate with the FDA or other relevant authorities to ensure you’re aware of upcoming changes.
5. Cross-Departmental Involvement
➸ What it is:
Labeling compliance often involves multiple departments, including regulatory affairs, quality assurance, and manufacturing.
Ensuring alignment across these teams is essential for successful labeling compliance.
➸ Why it's important:
Labeling errors can occur when different departments are not aligned on the final labeling specifications or when updates aren’t communicated effectively.
➸ How to do it:
Organize regular meetings between relevant departments to review labeling requirements and ensure that everyone is on the same page.
Create clear workflows to handle changes, approvals, and documentation between teams.
6. Quality Control in Label Design and Printing
➸ What it is:
Implementing quality control checks during the design and printing stages ensures that labels are error-free before being applied to products.
➸ Why it's important:
Even small label mistakes, like incorrect dosages, missing warnings, or misidentification of ingredients, can lead to serious consequences, including patient harm or regulatory action.
➸ How to do it:
Develop a checklist for quality control during the design and printing phases.
Pro tip
Signify enables you to evaluate every component of your product labeling, from ingredient listings and claims to warnings and formatting, ensuring they meet regulatory requirements across all markets.
With AI-generated checklists, Signify helps you identify gaps, track progress, and maintain compliance with industry standards, streamlining your labeling process.
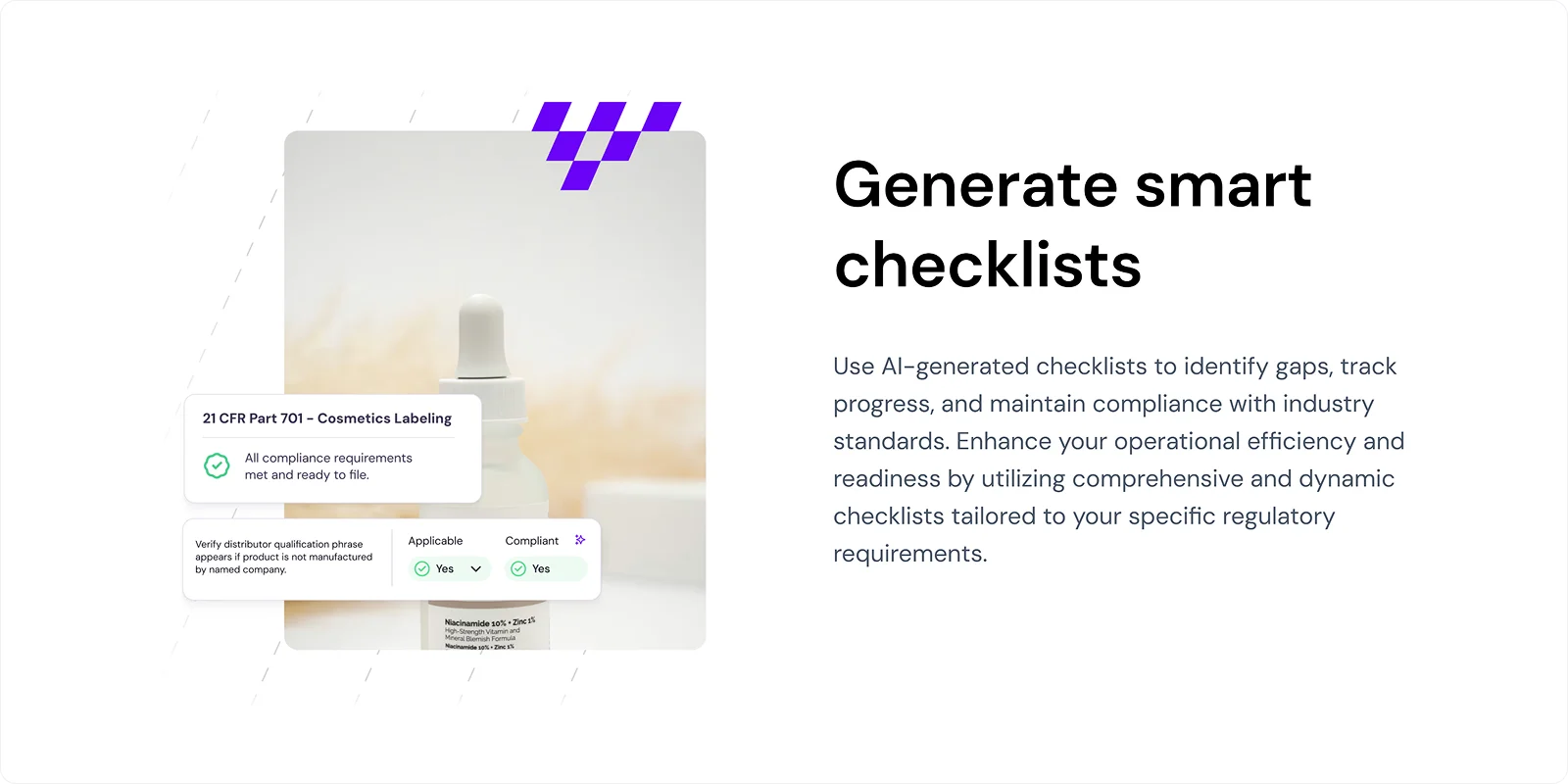
7. Traceability and Version Control
➸ What it is:
Maintaining a clear history of all label versions and the changes made to them, ensuring that you can track any updates or corrections made.
➸ Why it's important:
Traceability ensures that you can identify the root cause of any issues with the label (e.g., during a recall) and make necessary adjustments. It also provides documentation for regulatory inspections.
➸ How to do it:
Use version control software to manage label iterations and keep detailed records of changes and the reasons for each update.
For example, AI-powered systems can improve this by automatically creating version histories and ensuring that only approved versions are circulated.
Pro tip
Signify automatically documents every compliance assessment, providing clear records of who made decisions, what was evaluated, and when changes occurred, ensuring complete traceability.
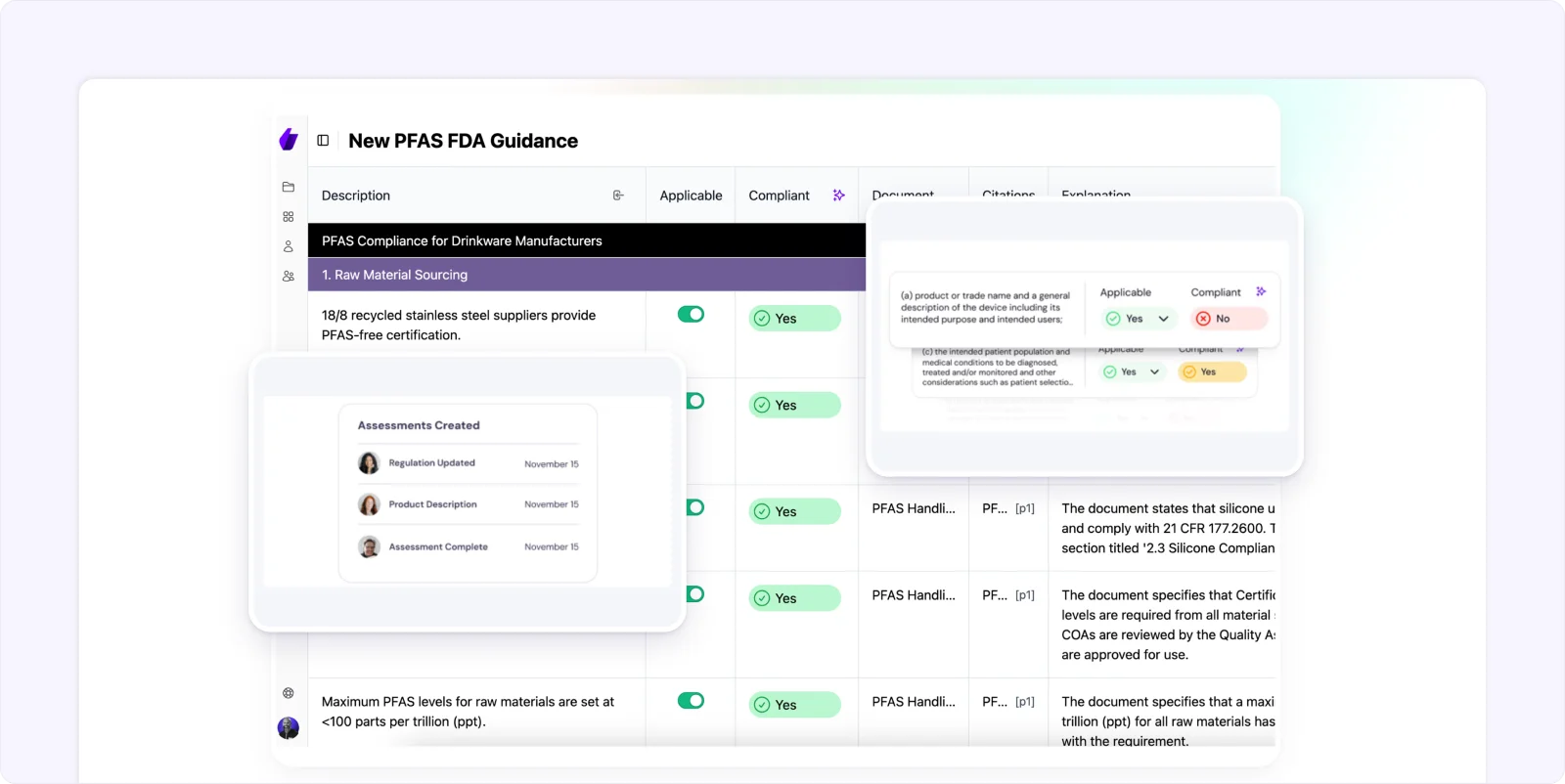
With comprehensive audit trails, you can prove compliance diligence by demonstrating due diligence to regulators and maintaining clear accountability for all compliance activities, making it easier to defend your compliance posture during audits.
Signify: A Leading Platform for Pharma Labeling Compliance
Signify is an AI-powered compliance automation platform that simplifies pharmaceutical labeling processes, ensuring that your products meet regulatory standards and remain compliant with industry guidelines.
By automating key tasks and providing real-time insights, Signify helps pharmaceutical companies stay on top of regulatory changes, reduce the risk of non-compliance, and streamline their labeling workflows.
Key Benefits of Using Signify for Pharma Labeling Compliance:
Proactive Compliance Monitoring: AI-driven analysis reviews product labeling and documentation to detect regulatory inconsistencies early, preventing potential compliance issues before they arise.
Smart Documentation Management: Automatically organizes and links labeling records, ensuring easy access and audit readiness while maintaining clear traceability for reporting and inspections.
Regulatory Risk Intelligence: Monitors real-time regulatory updates, alerting businesses to emerging risks so they can address labeling discrepancies before they become compliance concerns.
Guided Remediation Actions: Offers clear, actionable recommendations to resolve compliance gaps, ensuring that your labeling practices stay aligned with ever-evolving regulations.
Signify also serves industries beyond OTC Healthcare, including:
Food & Beverage
Personal Care & Cosmetics
Household Goods
Pet Care
Tobacco
Consumer Electronics & Appliances
Furniture
Tools & DIY Equipment
Sporting Goods
Ready to streamline your labeling compliance?
Book a demo to discover how Signify automates your pharma labeling workflows, mitigates compliance risks, and ensures your labeling processes are always up to date.


